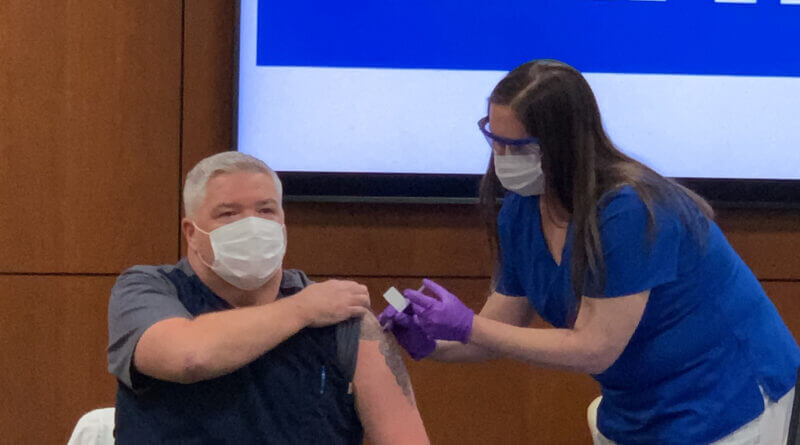
First COVID vaccines issued at Baptist-DeSoto
The most important weapon to date in the war against COVID-19 has found its way to the battlefield of DeSoto County. Wednesday, it was issued for the first time to frontline warriors on that battlefield.
Doctors and nurses who specifically deal with COVID-19 patients at Baptist Memorial Hospital-DeSoto in Southaven were the first to receive the Pfizer-BioNTech coronavirus vaccine. The doses are the initial shots of 975 vaccine doses allotted to the Southaven hospital.
Those who received the vaccine are part of the initial phase of the COVID-19 vaccination program, which includes health care professionals and long-term care facility residents.
Brian Hogan, Baptist-DeSoto Administrator and CEO, said the initial shots given are specifically for those doctors and nurses who directly care for COVID patients at the hospital on a nearly daily basis.

“We encourage people to take it and we are hoping that most of our employees will take the vaccine,” Hogan said. “Employees with direct care of patients are eligible for the shot and we’ve had a really good turnout of people signing up already to take the vaccine.”
Dr. David Hooker, ER Medical Director at Baptist-DeSoto, was among the first three people to simultaneously receive the vaccine, and said he wanted to show the vaccine was safe and should be taken by as many who are able to.
“I wanted to help us move forward in the fight against this pandemic to help show that it is a safe process that all of our frontline heroes can partake in as well as the rest of America when it becomes available,” Hooker said.
The Pfizer-BioNTech vaccine is given in two doses with the second or followup dose administered three weeks after the initial dose.
Baptist-DeSoto is the third hospital of five in the Baptist Memorial Healthcare system to receive the vaccine this week.
The first was the NEA-Baptist hospital in Jonesboro, Arkansas on Monday, Dec. 14. The Mississippi Baptist Medical Center in Jackson was second on Tuesday, Dec. 15.
Baptist-Memphis and Baptist-Crittendon in West Memphis, Arkansas, will get the vaccine on Thursday, Dec. 17.
No specific date for vaccine distribution has been announced for the Methodist hospital system, including Methodist-Olive Branch, but it is expected that it will happen soon.

Hogan said the vaccination distribution will be done in levels depending on their work at the hospital.
“The Level 1A, which we are in now, are those who on a consistent basis take direct care of COVID patients,” Hogan explained. “Then, 2A are next and they may be pharmacy staff or radiology staff who are very important to the hospital, but they may not take direct care of a COVID patient every day. Level 3 would be all other employees, such as administration, business office staff, people that are here around the COVID patients but we don’t take direct care of them.”
The priorities for who gets the vaccinations was determined by the Advisory Committee on Immunization Practices on Dec. 1. The Pfizer-BioNTech was approved for use on Dec. 11 on an emergency use authorization.
The general belief is that it may not be available for general use until summer, but Hogan said Baptist does have a plan in place to address that when the time comes.
“The first wave is just for hospitals and long-term care facilities and those people taking direct care of COVID patients,” Hogan explained. “This will be government regulated but as more vaccines are available they will open it up in phases on who can order the vaccine. Now, only hospitals and long-term care facilities are able to order but as soon as they release the instructions it will be prioritized on who falls next that can begin ordering and receiving the vaccine.”
Those employees who are getting the vaccinations are aware there will likely be a mild reaction to the vaccine for the first day or so after taking the shot.
“Part of the education that we give, and this comes straight from the manufacturer, is that they should or could expect some mild COVID-like symptoms for the day or two after the injection,” said Hogan. “It’s usually muscle aches, and maybe a headache or a low-grade fever.”
Hooker said he was ready for that because he felt the vaccine is an important step toward ending the pandemic.
“I’ve read the studies about the vaccines that are coming out and I’ve found that it is efficacious and is going to help everyone be protected against symptomatic disease and it’s got a really good safety protocol,” Hooker said.
The big question right now is how long is the effectiveness of the COVID-19 vaccination. Hogan said no one really knows for sure but we will learn that answer soon enough.
“The studies were done quickly so they haven’t given us a specific answer on how long the immunity will last,” Hogan said. “They have said it works but we don’t know for how long at this point.”
The Pfizer-BioNTech vaccine is rated as 95-percent effective. Hogan said Wednesday was a first step in moving the “new normal” back to “normal.”
“We think this brings us hope that we will be seeing the end of the pandemic soon,” said Hogan.